posted by Dave Arnold

Nils and I wrote an article on freezing for the September 2009 issue of Food Arts magazine. The piece included a short section on liquid nitrogen (LN or LN2), and I have greatly expanded it here for the blog. Important: we make no claims to inventing any of the techniques presented here. They are all pretty standard. Please, discuss who-came-up-with-what-first somewhere else. This is simply a primer.
Sections:
I. Introduction
II. Safety (very important stuff)
III. Getting and Storing LN
IV. Some Applications (cool stuff, most of the pictures)
I. Introduction:
Liquid nitrogen is about as cold as you can get in the kitchen, registering a whopping negative 196 degrees Celsius (-321° F) … and it’s non-diluting and non-contaminating to boot. Despite its preposterous coldness, liquid nitrogen has only 15% more cooling power than the same amount of ice at 0° Celsius. This counterintuitive fact leads many chefs to underestimate the amount of liquid nitrogen they need for a given task, like making ice cream.
Ice cream is typically the first thing people make with LN. Theoretically, it should be the best you can make because the mix freezes so quickly—and the quicker the freeze, the smaller the ice crystals, and the smoother the ice cream. But commercial ice cream machines make sufficiently small ice crystals for most palates, and it’s very easy to over-freeze portions of liquid nitrogen ice cream. So your LN ice cream results might not be as great as you expect.
But there are many other fantastic LN applications. You can turn fresh herbs into powder, separate citrus fruits and raspberries into jewel-like pieces, and freeze alcohol to make liquid centered treats. Be warned: liquid nitrogen is addictive and mesmerizing in the kitchen.
II. Safety:
Nitrogen makes up most of air we breathe. It is completely and utterly non-toxic—end of story. Yes, it is a chemical. Cows and carrots are just collections of chemicals.
The dangers of liquid nitrogen, of which you must be ever vigilant, are cold-burns, asphyxiation, and pressure-related explosions.
Dangers to Your Customers:
Don’t serve items to your customers too cold. You will burn their tongue, spoil their meal, and make them angry. I am speaking from first hand experience here (as the burnee, not the burner).
Dangers to You, Your Co-Workers, and Your Building:
Cold Burns:

If a small amount of liquid nitrogen touches your skin and rolls off, you will not be harmed. Lab technicians routinely dip their bare hands directly into liquid nitrogen (quickly! they don’t hold their hands in it). A layer of vapor instantly forms and temporarily protects your skin—this phenomenon is called the Leidenfrost Effect. Read a cool paper on the Leidenfrost Effect, written by a science teacher who dips his hand in molten lead here.
Burn danger occurs when liquid nitrogen remains in contact with your skin—whereupon liquid nitrogen burns the same way hot oil does. Painfully.
• Do Not wear clothing that can capture liquid nitrogen run-offs or spills, such as cuffed pants. I got my worst LN burn when my protective sleeve (which was not rated for cryogenic work—wrong choice) got too cold, cracked, and allowed LN to seep into the cloth cuff of my gloves.
• Do Not dispense LN into a container, such as a bain marie, that you are holding with your bare hands. You will get burned if the container gets super-chilled, which is likely. This hurts, too.
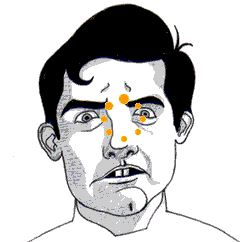
• Always wear safety goggles—LN can boil up and into your face at any moment, quickly blinding you. Forever.
• Never pour LN above your head, and do not kneel next to someone pouring LN.
• Do Not hold containers of LN with one hand, or in any other precarious way.
• Have a clear exit path in case something goes wrong—if you drop a whole container of LN, you and everyone else around you needs to get away from it quickly.
Explosion:
Never seal liquid nitrogen in a closed container. The pressure will rise, and unless the container can hold 1000 psi or better (nothing you have can) it will explode. In July 2009 a young cook in Germany lost both his hands and landed in a coma when he sealed LN in a closed container. Liquid nitrogen containers, called dewars, are either completely vented to the atmosphere or held between 10 and 22 psi with multiple safety valves. It’s worth repeating: don’t put LN in a container that is sealed or could become sealed by mistake.
• Let’s put that last part another way: Make sure there is no way your containers could become sealed. Example: a venting tube in a lid could get clogged, or crimped. A thermos lid you leave unscrewed can be sealed by your colleague. Note that even if an explosion isn’t strong enough to cause damage, LN spraying all over the place is hazardous.
• Never send LN through a pipe that could become sealed. You’ll make a pipe bomb.
• Never modify a dewar. In 2006 at Texas A&M, someone intentionally defeated the safety valves on a LN storage dewar. The resulting explosion destroyed several rooms, tore a hole in the ceiling, and ruptured plumbing that in turn flooded the entire building. Thankfully all this happened at 3am, and no one was hurt. The pdf report complete with pictures is: here.
Asphyxiation:

A small amount of liquid nitrogen turns into a large amount of nitrogen gas—LN expands by 700 times when it vaporizes. If you use a lot of LN in a closed space, it will displace the oxygen and suffocate you before you know what’s happening. This is the largest cause of death from LN. The danger is very, very real.
Unless you have been trained like an air force pilot—by coming up to the very brink of nitrogen-induced death under highly controlled circumstances—you can’t tell that you are suffocating on nitrogen. The panicky feeling you get from choking or staying under the water too long doesn’t come from lack of oxygen— it comes from the buildup of CO2 in your blood. Your lungs can get rid of CO2 just fine in a pure nitrogen environment, so your body doesn’t send those helpful distress signals. Also, breathing pure nitrogen is much worse than just holding your breath. Breathing nitrogen actually sucks the existing oxygen out of your bloodstream. Majorly bad news. So,
• Do Not use LN in an unventilated area. What constitutes an unventilated area? Read ASU’s guidelines here.
• Never go into an elevator with LN. If a dewar breaks in the elevator, you’re dead.
• Never carry LN in a vehicle’s passenger compartment. You get into an accident, the LN spills and vaporizes, you suffocate instantly.
• If you see someone passed out near an LN tank, do not try to help. Many people who die from nitrogen suffocation were trying to help co-workers. If the victim has passed out they are probably already dead. Call 911.
• Invest in an oxygen meter, like the one shown in the picture above.
• We can’t cover every eventuality here—be sensible.
Set up and enforce a safety program:
Make sure everyone who uses LN is properly trained. Print out and post basic LN safety rules. Some documents:
An overview of the hazards (plus gruesome details) from the US Chemical Safety Board.
The cryogenic fluids handling guide from Arizona State University.
III. Getting and Storing LN:
Unlike compressed gasses, liquid nitrogen isn’t stored under high pressure. In a cylinder of nitrogen gas, the pressure can easily top 1000 psi. It can sit around at room temperature practically forever without any loss. LN, on the other hand, is always freezing cold and is kept in an insulated container (called a dewar) under little, if any, pressure. Because LN is constantly boiling off, you’re always losing a little bit. The bigger and better insulated your dewar, the longer you can store your LN. Because LN is always boiling off and venting through those all-important relief valves, your dewar will occasionally makes little psssssssssssssssss sounds. Don’t be alarmed by the hissing noise—it’s your friend.
When you are filling a container or dewar remember this: you will use up a lot of LN just chilling down the vessel. Doesn’t matter if you are filling a dewar, a bain, or a Styrofoam cooler—when you first add LN, it will smoke furiously while the surface of the container gets chilled down to LN temperatures. After the container is chilled, it will fill up in an orderly and efficient way. Rule of thumb for LN efficiency: don’t let your containers run dry and warm up if you don’t have to. Keep them cold.
The easiest way to get LN is to snag some from a buddy who has a bunch already. Use a vented vacuum insulated coffee dispenser, never a thermos! A thermos can seal and explode. Many vacuum insulated coffee pots are inherently vented. Check to make sure yours is.

We use these pots quite a bit around the school. They are convenient, easy to pour from, and they only lose about 50 percent of their volume in 24 hours. I’ve heard people tell of getting these pots filled at their local gas supplier, but I have had no such luck.
Most chefs obtain liquid nitrogen from compressed gas/welding suppliers. You’ll have two choices: buy or rent a small 10-50 liter capacity dewar and have it filled on a regular basis (few places in New York still rent these dewars), or buy or rent a large 160-240 liter capacity storage dewar and have the company swap your empties for fulls when necessary. The large dewars are much more economical; up-front costs are higher ($2000 versus $1000) but they’re much cheaper to own. You’ll pay the same amount to swap out a 160 liter dewar as you will to fill a 50 liter dewar. If a small dewar develops a leak or breaks, it’s your problem—not an issue with large dewars because they are swapped out. You can usually rent the large dewars for a small monthly fee, plus the rather large refundable deposit. One annoyance with the large dewars: you will need to prove to the supplier that you own it. They won’t swap out your dewar if they think it’s a rental from a competitor or stolen off the street from the local utility company.
Small Dewars, 10-50 Liters:
10 liter dewars are small and convenient, but they don’t have nearly the holding time of larger dewars. Remember—the larger the dewar, the smaller the percentage of LN that evaporates per day. If you have to pay someone to come with a truck and fill your dewar, 10 liter dewars are uneconomical—go for 35-50 liters.

A 10-20 liter dewar really shines if you need to carry LN from place to place, or store the last of the LN from your big dewar while it’s out being refilled. Hint: you’ll want to order LN before you run completely dry, but you don’t want to throw away the last of your LN. Save the dregs with a 10- or 20-liter plug top dewar.
Liquid Withdrawal Device:
Dewars with liquid withdrawal devices will cost you more, but they’re worth it for their convenience. Here’s an image from dewar manufacturer Taylor Wharton:

The liquid withdrawal device just clamps onto an ordinary plug-top dewar. The clamp seals the top and allows the dewar to develop about 11 psi. Above that pressure, excess nitrogen is vented from a relief valve— which you will hear hissing. Sometimes, if the dewar is overfilled, the valve will get frozen open and you will lose all your LN. Try tapping the valve to shut it or gently heating it to unfreeze it. If the relief valve clogs and fails (as opposed to freezing open, which is not a safety issue, but a money issue), the secondary safety valve kicks in. Here is the operating manual for the liquid withdrawal device, including instructions on how to fill a dewar.
Big Dewars:
If you really want to be an LN ninja—and save money in the long run—buy a real dewar—160-240 liters. Here are the parts:

The dewar guys will try to sell you a pricey, easy-to-break liquid withdrawal hose. Forget them, and build your own liquid takeoff. All large dewars have a ½ inch flare connector on the liquid valve. Here are the parts you’ll need:

Notice our inexpensive “cryogenic phase separator.†Don’t skip this part. Take a look at this dewar opened up with, and then without, the muffler:

Here is the owner’s manual for the large dewars.
IV. Some Applications—the Fun Part:

Remember: these are basic techniques. No one is taking any credit. See the first paragraph of this primer.
Ice Cream:
LN ice cream is typically made in a Kitchen-Aid with a paddle attachment. Don’t use the whisk. All the mixer is really doing is stirring the LN into the mixture, not providing aeration. LN boils violently, so it provides both aeration and freezing. But the LN also floats—so without the mixer you will use a ton of LN and just freeze the surface. A liter of ice cream base at refrigerator temperature will take roughly a liter of LN to make ice cream. Turn the mixer on medium speed and add the LN slowly to prevent splashing. LN fog will be everywhere, and you will see nothing. Wait for the fog to disperse. Scrape down the sides of the bowl with a stiff metal spatula. If the mix is too soft, add more LN. If the mix is too hard or has hard lumps mixed with soft parts, turn the mixer on high for a bit to even out the texture.

Chilling Drinks and Other Liquids:
LN is a fabulous way to chill liquids without diluting them. Here is the simple rule: always add your liquid to LN rather than the other way around. LN floats. If you pour LN on top of a drink you will get a frozen layer on top with a warm layer on the bottom, and will use far more LN than necessary.

It doesn’t make sense to chill individual drinks with LN. It is very easy to overshoot when chilling small quantities, and you’ll freeze your liquid solid. LN is a great way to chill a lot of drinks—it’s as fast to chill 100 as 1. If you freeze your drink too solid, the heat from your hands, a torch, or hot water on the outside of the container can bring it back. Alcoholic drinks melt pretty quickly.
So How Frozen Should I Make My Drinks?
You need to be sure you aren’t going to freeze any tongues. Just because you can freeze vodka solid doesn’t mean you should. Straight liquor shouldn’t be served below -20°C (-18°C is better, a -20°C drink has a sting to it). The higher the water content, the more cold punch it will pack. A -10°C drink at 17% alcohol will taste a lot colder than a -10°C drink at 40% alcohol. Here is a visual guide:

By the way: you can use LN to freeze Pacojet containers in under a half hour, including tempering—but I shouldn’t print the technique because I’m sure it will void your warranty, and I don’t want all those broken Pacojet blades on my conscience.
Chilling Glasses:
LN is the best way to chill glasses, and it costs less than 10 cents a glass. I love this technique. The inside of the glass is chilled, but the stem and the base remain condensation-free and the outside of the glass doesn’t get so cold that your lips stick. To look badass, chill at least two glasses, one in each hand, simultaneously.

This technique works well with champagne flutes, highballs, rocks glasses, etc. Don’t use this trick with a martini glass; as you swirl you will send LN spraying into your face.
Blowing Smoke:
You cannot eat high-moisture foods right when they come out of LN. They will freeze to your tongue and cause you great pain. Low moisture foods—like dry meringues, marshmallows, some cookies, some cakes—don’t transfer a lot of energy from your tongue, so you can eat them almost right out of the LN. Added bonus: when you crunch these foods in your mouth, a plume of scented (and harmless) nitrogen fog shoots out of your nose and mouth.
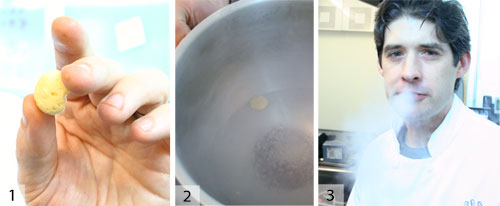
Breaking Things Up: Raspberries, Citrus, and Powdered Fresh Herbs:
Things frozen in LN tend to get brittle. Use this fact to your advantage, as in the following three techniques:




Freezing the Un-freezable and Freezing in Cool Shapes:
Though this article is not about history and credit-bestowing, I have to make an exception here because Dani Garcia, of El Restaurante Calima in Spain, effectively owns the olive-oil process I’m about to explain. He has used LN to freeze olive oil and olive oil emulsions into more shapes and textures than you can imagine. LN-frozen oil doesn’t hurt your tongue because the oil doesn’t pack the heat wallop that water does.

The last technique I’ll show you is currently on the menu of our friend, Johnny Iuzzini of Restaurant Jean Georges. He makes chocolate squiggles and branches by piping chocolate into LN with a cornet.

This is a variation on the main idea that LN can make anything stay put by freezing it quickly. The possibilities are endless.
Let’s wrap this up. LN’s going to hook you quick. I remember seeing some Spanish chefs do a demo in 2004. I noted to myself that they used liquid nitrogen like it was water, whereas I was rationing it like wine. Now that I have the big tank, I rip though it just like they do. And you will too.
Even — or especially — as someone who will probably never work with liquid nitrogen, this was fascinating. Thank you.
wow thats amazing… I have been waiting for this post for such a long time…now cant wait to get my hands with some LN2….
Great primer!
I avoid plastic bowls and use metal ones when playing with liquid N2 because I am afraid plastic might fracture. Not sure if it is neccesary though.
My favorite:
If you have a Dewar with wide opening like shown above you can “cold fry” whipped cream. Squirt directly into the nitrogen and use a wooden spoon to push the islands under. You will end up with reverse ice cream with whipped cream.
Our lab uses 200-400 liters of liquid N2 a week so in summer I play around with it for parties etc.
Howdy Jurgen,
Nice. I think you are correct about the plastic. We don’t use it either. It is in the pictures just so people could see what is going on. The polycarb seemed ok for the shoot but I was worried it would crack. Quart containers and acrylic almost always crack. Heck, I’ve even had 304 stainless bains crack.
This is so awesome! I hope someday i’ll get to work with LN. No chance that this is something I can look forward to in Pastry III at the FCI, is there Dave? If you ever need an intern, please email me!
Stop by.
When chilling beverages, how will you separate the drink from the liquid N2 once the desired temperature has been reached?
Hi Derek,
That’s the problem. If you don’t add the correct amount of LN at the get-go you end up overfreezing your drink. Unlike dry ice, where there is the danger of serving someone chunks of dry ice by mistake, LN almost always keeps fogging so long as you stir it till it is completely gone.
how is liquid nitrogen that you buy at hydroponic garden suppliers different that liquid nitrogen you deal with here ? the nitrogen i have in front of me is neither cold or in a un-vented container but perhaps in some water solution ?
Howdy J Walker,
I’m not 100% familiar with the product you have, but I believe the hydroponic stuff is nitrogen containing compounds (like ammonium nitrate) in a water base. These things exist fine at normal temperatures. Liquified Nitrogen, on the other hand, is pure N2 that has been cooled to the point that it changes from a gas to a liquid (a couple degrees colder and you can actually turn it to a solid. you can do that in a vacuum machine). Hope this makes sense.
Hey Dave
Great stuff i did the Harold Mcgee series November 08 and still using the reference material to this day.
Dave: Great article.
I’ve been running a large weekend party based around ice cream making for 20 years, using LN2 for the ice cream since ’94.
Years of experience say that 5 gallon plastic food prep buckets (and cheap-ass 1 gallon pails) will take endless batches of LN2 Ice Cream just fine.
We’ve never actually spent the money for polycarb prep containers as it’s never been necessary.
We decided against metal for the mixing as you do spend a lot more LN2 cooling the metal.
http://www.flickr.com/photos/fabulous_redhead/3721373930/in/set-72157621456675818/ shows our standard batch production method (the small dewer is for easier handling. We fill it from the 200 that’s out of the way, off camera), and http://picasaweb.google.com/vvalkyri/Baitconxx02?authkey=Gv1sRgCMirguLsoNSO0gE&feat=email#5355029073894043426 is me, about 2 minutes after having a couple of litres of LN2 dumped on my head. The only problem I’ve ever had has been, in fact, when the LN2 gets caught somehow – say in the sleeves of my shirt… but it was only minor frostbite. 🙂
Regarding the powdered spice: Have you tried putting the frozen powder under a stiff vacuum while it thaws? It seems like this might be a great way to have super fresh dried spices.
Howdy Greg,
That might work if you put a cold trap in the vacuum line. I’m afraid you’d lose a lot of the volatiles though.
Hiya Dave,
I was sloppy earlier; I should have been saying ‘herbs’.
My thought was something along the lines of freeze drying the herbs so that you’d end up with some room temp stable fresh herb powder.
My process would be to prepare everything up to plating, but instead of going onto a plate, the herbs would go under vacuum to remove the water. (I was also, perhaps naively, thinking that the vacuum pump would remove water vapor.) I was thinking that sublimation wouldn’t lead to the loss of water soluble volatiles, but now I’m not sure.
So say it does. Could you throw in some amount of a hydrophilic ingredient that also works with a dish? Depending on the dish, maybe sugar, flour or cornstarch. The key would be to keep them separated so you could play around with the mix. This might also help test the volatile losses.
It seems like there is a potential that this could be some sort of rapid freeze drying process.
Hi dave i already took your sous vide class, the last february, i need to ask you some questions, first how long should you cook a rack of lamb to medium and at what temperature y got 300 gr new zeland lambs so i dont want to spent most of it trying , , and second dave i want to buy liquid nitrogen but they told me the what 2 clases here in south america , what is the name ans specifications of the nitro used for food, please help me, and well thank you very much for your answers Ovidio Gabela
Hi Ovidio,
Regarding nitrogen, I don’t know of differnet grades of liquid nitrogen. Perhaps there is a hospital grade, but I’m not sure. There are two grades of nitrous (N2O). For that the car grade is poison. You need the USP grade.
Regarding Lamb,
The lamb shoudn’t take more than an hour to cook (but it won’t be pasteurized). I have never cooked one to medium. We usually cook to 56 C, cool to 52-54 C and finish sear.
I have a bottle of liquid Oxygen, left over from a home breathing apparatus. It’s even colder than Nitrogen, so I’m going to use that. I’ll just drill a hole into the tank above the level of the liquid and tap in that way.
I can’t wait to try some of these techniques out!
Bob,
NOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOO! Liquid Oxygen (LOX) is extremely dangerous (it is also actually not quite as cold as LN). Do a web search on LOX BBQ. Also, don’t ever, ever, drill into a pressurized tank. Ever. Even if it is unpressurized, never drill into a tank containing a potent oxidizer. I hope you were kidding.
Dave,
Thanks for the great ideas and safety tips!
How much should you normally expect to pay for LN2? I’ve seen websites that make claims that it costs less then beer or milk, yet the local supplier wants $3.74/L for medical-grade LN2. Does this sound like a reasonable price?
Thanks!
Hi James H,
That is a reasonable price for a small amount, but horrendous for a large amount. I have a 160 liter dewar. It costs $125 to swap out. A 180 liter dewar costs the same. A 240 liter dewar costs like 150-175. Here is the kicker — a 30 liter dewar costs around $75 to fill up.
Hey Dave, I’m shopping for a LN2 Tank Ninja level. Could you look at this one and advice me if it’s good… http://www.labx.com/v2/spiderdealer2/vistaSearchDetails.cfm?LVid=6993534
Also, I saw some on ebay that are being sold “as-is” and are “working pulls” do you have any advice with buying these units? If the unit I get is not working could I just swap it out?
Thanks, any advice will be sweet as I’m looking to invest in a Ninja Level LN2 tank.
Hi Tali,
How much do they want for it? The trick is gwtting your local welding supply to swap it for you. I’d figure out the price, make sure your welding supplier will swap it for you, and then buy it if it is less than 1000 bucks.
Great and very complete post!
Liquid nitrogen is some very fascinating stuff. I usually like to think about it as a reverse hot oil, you can do more or less the same things and use many similar techniques.
Too bad one lives in Italy at the moment though – have a look at this!
http://www.caputmundicibus.com/2010/02/italian-government-legislates-against-molecular-cuisine/
/ john
Hi John,
I just read you post. Is that for real? It doesn’t make any sense. Agar is a traditional ingredient, as is carrageenen, as is Locust Bean Gum, etc, etc etc. Are they banning sugar? Surely they aren’t banning gelato stabilizers. All of Italy would go haywire!
I know, it’s absolutely crazy! Problem is that Gov’t & people do not know what these “powders” are and therfore take a hostile stance towards it instantly. All people here know about Molecular cuisine is – “chemicals” – “oh, it HAS to be bad for you then…”
Traditional fear of progress 🙁
Hi, great info! I am looking at buying a 10L dewar, but unsure of how much nitrogen 10 liters really is. How much do you typically use per session? Would 10 liters last a few hours? Can you give me an idea of the rate it evaporates, or what your usage typically is.
I’ve found a place locally that will fill whatever size dewar for .75 per liter and will rent a 160L for $30 a month. I just want to check this all out before committing to a year lease on the jumbo tank.
http://www.intlcryo.com/popups/dewar.htm
I’m looking at the IC-10D
Thanks
Chad
Hi Chad,
The best use for the 10 liter dewar is moving LN around, having at the service line, and to store the last bit of LN from your big dewar when the company swaps them out. I wish I had one for that –I’m always losing money by giving the welding company the last of my LN in the dewar back to them. Once you have LN and start using it, it would be difficult to deal with having only 10 liters. You will be in constant fear of running out. Evaporation in the 10 liter dewar is low –but nowhere near as low as the 160. If you have the space, I’d go for the 180 or 240. They don’t usually cost more to rent.
This is quite possibly my new favorite blog. Not only do you guys play with LN2 and other awesome food-related stuff, you buy things from McMaster-Carr, my favorite company ever. We use those sintered mufflers for our LN2 dewars also (I’m a physicist; in our lab we work with both LN2 and liquid helium).
You ever look into producing your own liquid nitrogen? I was just reading this, and measuring my spare room:
http://benkrasnow.blogspot.com/2008/08/diy-liquid-nitrogen-generator.html
Likely like the output isn’t enough for your needs, but the idea is fun.
Pretty ballsy
Dave,
Really nice post, its complete and the bonus of some techniques is a +, cool that you take a good part in safety.
I’m looking to get LN into the dessert. I don’t know if you have heard about Burning Man, it takes place in Black Rock City, Nevada. During the day temp can rise up to 104f and by night can drop to 30f. The problem is that I have to get the tank into a truck in San Francisco and make all the way up to BRC, is a 10 hours drive. I’m planing to use it as soon as it get there in Cuba Libre frozen popsicles.
Appreciate your thoughts in this matter.
Pickup truck?
Just want to ask a few questions.
1. How much volume of liquid nitrogen will be needed for 1 serving of frozen popcornsicle?
2. Could I store the dewar at room temperature and where in the kitchen?
3. How do I transport the dewar? could I put it in my car?
4. Could I pour liquid nitrogen in plastic cups?
Thank you!
Hello Oliver,
4. Plastic cups will most likely shatter when filled with LN –be careful.
3. I would not put a dewar in the car. Back of a pickup truck is perfect.
2. You can store a dewar in you kitchen if it is very well ventilated.
1. Figure equal volumes of LN and product. If you are freezing something really small you’ll need more because of losses.
Is it OK to bring a small dewar of LN in an elevator? How else do you get it up to a high floor? Do you put it in an empty elevator and send it up? Thanks!
Nick –How small? If the elevator crapped out and all the LN spilled on the floor, would you die? That is the only real criteria you are dealing with. 3 liters in a large elevator won’t kill you. 50 might. I put LN in empty elevators and send them up posting a sentry at each floor to make sure no-one gets on. Pain in the butt but better safe.
I recently had ice cream from a shop that specializes in LN freezing and it was way too sweet. Am I correct in assuming they over-sweetened the batch in order to keep it from freezing too hard?
P.S. Love your blog–sounds like you’re really having fun!
Interesting question Catherine,
It is definitely possible to get LN ice cream that is over sugared nice and hard (which would be more difficult using a normal machine), the same way you can make alcohol into a “sorbet” (but it is often not a good idea because it is just too darn cold). There is no reason to over sugar your base just because you use LN. Maybe it helps them –I haven’t tried it. The weird thing about LN is if you over-freeze your ice cream you will get pockets that are too hard, and pockets that are right (lumps), but all the ice cream is OK. If you temper it, the texture will even out.
How do you prevent the 100 decible noise that the dewar makes when it releases pressure out its valve? As this will drive away customers.
Dewers aren’t that loud when we use them. They are only pressurized to about 22 psi. The outlet is capped with a “phase transition device” (really a sintered bronze muffler). We use it all the time in front of people. Now… cracking the valve on an 800 psi CO2 tank is another story.
I just found a local welding supply place that sells liquid nitrogen by the pound. I am really excited about starting to work with the substance, but won’t be able to get a dewar. They are just too expensive. I called the welding supply and asked for suggestions. They said they have people come in with plastic coolers (although they did point out that the interior plastic does crack), animal semen containers and dewars.
Given my situation and the fact that I won’t need a lot of liquid nitrogen at a time (cooking for 2) do you have a suggestion on what to use? It seems a small plastic cooler would be the least expensive option, but seems like it might not be safe? Do you have any thoughts or alternative suggestions?
Howdy Andrew,
The best containers are the study styrofoam shipping cubes that are used to ship tings in dry ice. Find one with roughly the volume you want then make sure it is protected by a sturdy, close fitting, cardboard box. Plastic coolers can crack (although I’ve done it many times) and the disposable styro coolers from the supermarket break too easily.
Hi, Dave-
I would like to give you a device for easier handling of ln2. Please check your gmail for a message titled, “liquid nitrogen”
Is dealing with LN2 practical for small-time home use? I just want a small amount for making a few batches of ice cream with some friends for one afternoon and don’t want to deal with big 100L+ dewars. Is that viable? Or do vendors not deal with small amounts?
Hey Alex, It depends on where you live. If you live in a big city it will probably be a PITA. If you live in a suburban or rural area, you might be able to convince the local welding shop to sell you some in a non-approved dewar.
Hello, I am planning on using LN for making alcoholic Popsicles for a 2 day festival any suggestions on how much I might need for it to last atleast 3 days. Also any technique suggestions would be helpful I was planning on making the mixtures and then pouring them into Popsicle molds (plastic?) And then submersing the molds in the LN. Has anyone else done anything like this?
I’d just rent a 160 or 180 liter dewar –it is much cheaper this way, you don’t have to worry about running out and the tank will hole for a long, long time. I’d be careful, cause the popsicle molds will become quite brittle. You’ll have to use silicone and let them temper back to normal after you make them.
I’ve rented a 230 liter tank and found stainless steel Popsicle molds, delivery this week, im.gonna try a few ideas, mostly im afraid of burning someone with them
Mark–that is exactly the thing to be worried about. How did it go?