I. What is TG?
II. TG Safety, Nutrition, and Taste
III. Getting TG and Types of TG
IV. Storing and Testing TG
V. Salt and TG
VI. TG and Cooked and Cured Meats and Fats
VII. Important Safety Note
VIII. Using TG
IX. A Note on Proteins
I. What is TG?

Transglutaminase (TG or TGase), better known to chefs as “Meat Glue,†has the amazing ability to bond protein-containing foods together. Raw meats bound with TG are often strong enough to be handled as if they were whole uncut muscles. TG is safe, natural, and easy to use. In the kitchen, TG is primarily used to:
• Make uniform portions that cook evenly, look good, and reduce waste
• Bind meat mixtures like sausages without casings
• Make novel meat combinations like lamb and scallops
• Produce special effects like meat noodles, meat and vegetable pastas (using gelatin as a binder), etc. Additionally, TG can thicken egg yolks, strengthen dough mixtures, thicken dairy systems, and increase yield in tofu production, among other useful applications.
This primer is aimed at giving the basics of meat glue—not recipes. Individual recipes abound on our blog and the net.
Â
Â
TG is a naturally occurring enzyme in plants, animals, and bacteria. Enzymes are proteins that act as catalysts in chemical reactions; they speed up reactions and make reactions occur that otherwise wouldn’t. Although TG is a newcomer to the kitchen, cooks have used enzymes for thousands of years. Enzymes in papaya, for instance, are traditionally used as meat tenderizers. The enzyme rennet is used to curdle milk when making cheese. Enzymes that break down starches into sugar are used to brew beer. Naturally occurring enzymes in meat tenderize dry-aged steaks and give cured meats their distinctive flavors. Enzymes are the reason fresh pineapples hurt your mouth. They also keep gelatin from setting.
Â
TG bonds protein molecules together with a very strong (covalent) bond by linking two amino acids: glutamine and lysine (for exactly what goes on see A Note on Proteins). TG was first identified in 1959 and isolated for testing and research in the 1960’s. Guinea pig liver provided the first commercially available form of TG. Though researched for food applications, TG was not widely used at the time because it was expensive, difficult to refine, and required calcium to work. In 1989, after testing over five thousands strains of microbes, researchers at the Japanese company Ajinomoto (best known for the production of MSG) discovered a strain of soil bacteria, Streptoverticillium mobaraense, which produced large quantities of easily purified TG. This TG is called microbial TG, or mTG. Besides being easy to produce, mTG does not require calcium and is very easy to use.
A Personal Note from Nils:
With meat glue you can glue any protein to any protein. Great, right? Well, just because you can do it, doesn’t mean you should. Gluing salmon to salmon is a very good idea. You get a piece of fish that is uniform; looks the same, cooks the same. Gluing chicken to salmon is not such a great idea because they have different textures and different cooking temperatures. The salmon will be dead before the chicken is cooked. Gluing chicken skin to salmon works quite well though, and will actually protect the outside of the salmon from overcooking. The other thing to take in consideration is how many pieces of meat you are fusing together. Some people try to make the case that you can use all the small pieces you have left over from fabricating to make a whole muscle cut. For example, the trimmings from tenderloin can be glued together to make a couple of extra portions –but they aren’t fillet mignon steaks. Bad idea. With too many small pieces, the meat will just look “Frankensteined.†Also, the texture will be completely different than a piece made from two or three (maximum) pieces.
II. TG Safety, Nutrition, and Taste
Short Story:
TG is safe. It will not harm you or glue your hands together. TG is deactivated by most cooking techniques and imparts no off-flavors to foods. (There is some debate about off-flavors. Read the next section, Long, Long Story.) TG is delivered as a powder and, like all powders, should not be inhaled. TG should not be consumed directly in large quantities, but consuming active TG in the levels recommended for food usage is harmless. TG is classified by the FDA as a GRAS product (generally recognized as safe) when used properly. Although some studies have shown that stomach enzymes have difficulty breaking down proteins after they have been bonded by TG, other studies have shown that these bonded proteins are absorbed and broken down in the body into normal products as though they had never been bonded.
Long, Long Story:
Taste First (We are a Cooking Blog!):
Normally, we detect no off-flavors in products bonded with TG. Bruno Goussault and Cuisine Solutions proposed that they could detect off flavors in long-cooked vacuum-packed beef products. We didn’t believe them so we tested it out. Turns out that a certain portion of the population can detect an off-flavor in short ribs that have been coated in excessive amounts of TG (Activa RM type) and cooked for 48 hours in a vacuum bag when tasted blind next to a control. If a moderate amount of meat glue is used fewer people can pick anything out, and if a light amount of meat glue is used, people detect an off-flavor in the control as often as the one with TG.
What is the reason for this? We aren’t sure. We have two hypotheses. The casein in Activa RM is causing the-off flavors (we are getting some Activa TI to conduct our own test). Or the enzyme itself is causing the off-flavors, either through the ammonia released during the reaction (maybe it can’t escape the vacuum bag? See our section on testing meat glue) or through some other reaction. The only other time I detected an off-flavor was in a raw fish product that had been heavily meat glued and vacuum bagged. That time I knew the culprit: the ammonia from the reaction itself. I lowered the amount of meat glue and cut the bag open to allow the product to air out before serving and had no further issues.
TG and Nutrition:
We have nothing more to say than what we said before: Although some studies have shown that stomach enzymes have difficulty breaking down proteins after they have been bonded by TG*, other studies have shown that these bonded proteins are absorbed and broken down in the body into normal products as though they had never been bonded**. In any event, even if the bonded proteins weren’t as bio-available as the native proteins, the actual percent of amino acids no longer available to the human body after digestion would only be a small portion of the total in the food.
*Influence of Transglutaminase-Induced Cross-Linking on In Vitro Digestability of Soy Protein Isolate, Chuan-He Tang, et al., Journal of Food Biochemistry 30 (2006), 718-731.
** The Epsilon(Gamma-Glutamyl)-Lysine moiety in cross linked casein is an available source of Lysine for Rats, K Seguro, et al., J. Nutr., 126 (1996), 2557-2562. We haven’t read this study.
TG and the Human Body:
There are many types of TG, including several in the human body. TG-ases play a vital and necessary role in skin synthesis, blood clotting, the immune response, etc. You wouldn’t exist without them. When TG-ases are improperly regulated in the body, they are associated with very bad things like the plaques in the brains of Alzheimer’s, Parkinson’s, and Huntington’s disease patients as well as in the development of cataracts in the eyes, arteriosclerosis (hardening of the arteries), various skin disorders, etc*. None of these are related to eating food made with mTG, but rather due to imbalances in the body’s ability to regulate the TG that it produces.
*Transglutaminases in Disease, Soo-Youl Kim, et al., Neurochemistry International 40 (2002), 85-103.
Potential Threats of mTG
The main threats theoretically posed by TG are that it is itself harmful or that the new proteins it creates are harmful. We’ll address each part separately.
Is mTG Itself Harmful?
Once mTG has been denatured it is a protein like any other that gets broken down in the digestive tract like any other. mTG is inactivated by heat. After 10 minutes at 60°C, 94% of mTG is denatured. mTG is eventually denatured even at 55°C. If you eat active mTG, it is inactivated by the low pH of your stomach and is broken down in under a minute by pepsin, one of your digestive enzymes. The only real hazards of ingesting active mTG therefore would occur in your mouth and esophagus.
The question is: Are there any hazards? In order to receive GRAS status from the FDA, Ajinomoto had to show that mTG was non-toxic. To do so they provided three basic arguments:
1.) Transglutaminase occurs naturally as has always been eaten to a certain degree.
2.) Rats fed large doses of Transglutaminase didn’t die or show any acute pathological problems.
3.) Different cell cultures that were tested for cell mutations using Transglutaminase showed no mutagenic effects. This test was to show that TG isn’t detrimental in the long term (Acute Toxicity and Genotoxicity Studies of a Microbial Transglutaminase, BK Bernard, et al, International Journal of Toxicology 1998. This study was paid for by Ajinomoto).
Ajjinomoto’s arguments have 3 holes in them that I can see. First, the argument that we have been eating TG for a long time is only partly true. We have been eating a different form of TG for a long time. MTG is a different enzyme from the one found in animal and plant tissue.  MTG doesn’t require calcium, binds more types of proteins, etc. Second, their study involves force feeding rats via a stomach tube. Once the enzyme is in the stomach it is quickly inactivated and destroyed. They don’t have any data on the acute effects of large doses of active mTG on the mouth, tongue and esophagus (that I can see).  Third, I’d like to see a long-term study of the effects of chronic ingestion of active mTG on an animal’s system. I wasn’t able to find any studies where rats were fed a ton of TG for 2 years and examined (I’m not saying these studies don’t exist). Even given those caveats, we are confident of the safety of TG itself. We have been eating it for years.
Are the new proteins that TG makes harmful?
The main hazard of creating novel proteins is their effect as a potential allergen. While TG itself is non-allergenic, there are studies that show that gluten can be made more allergenic if treated with TG. Gluten-free cereal products can contain a small amount of gluten. That small amount is below the threshold of activity for Celiac patients. If that small amount of gluten is made more allergenic by TG, there is the possibility that a “Gluten Free†product could suddenly pose a hazard*. Interestingly, there are other studies that claim that if gluten is reacted with mTG under the right circumstances, it can be rendered safe for consumption by Celiac sufferers**.
*(Microbial Transglutaminases Generate T Cell Stimulatory Epitopes Involved in Celiac Disease, E.H.A. Dekking, et al, Journal of Cereal Science 47 (2008), 339-346.
**Transamidation of Wheat Flour Inhibits the Response to Gliadin of Intestinal T Cells in Celiac Disease, Carmen Gianfrani, et al. Gastroenterology 133 (2007), 780-789.
III. Getting TG and Types of TG
Ajinomoto is the only producer of food grade TG, marketed under the brand name Activa (not Activia, which is a pro-biotic yogurt for women). Ajinomoto offers Activa to individuals in 1 pound increments. A kilo currently costs roughly $60 and will glue over 100 pounds of meat paste and a substantially larger amount of whole muscle pieces. Activa is also available through some online retailers. Activa is not pure TG, as the pure form is too concentrated to use easily. Instead, Ajinomoto blends TG with fillers (maltodextrin, a break-down product of starch) and other functional ingredients to suit the end needs of the user. The types available are:
- Activa RM:Most chefs use RM, which is designed to bond even problem foods like chicken breasts and cooked meats. RM is a mixture of TG, maltodextrin, and the helper protein sodium caseinate. Sodium caseinate is a water soluble protein derived from milk, and TG bonds it extremely well. The caseinate in Activa RM fills in the gaps between the pieces being glued, making up for any lack of available protein in the food itself. RM can be sprinkled on like a powder, mixed with 4 times its weight in water to make a slurry, or added directly into meat mixtures. Even if a recipe doesn’t require the extra bonding insurance caseinate provides, there is no disadvantage to having it mixed with the TG. For most applications, RM is the only TG you will ever need.
- Activa GB:  GB (“greatest bondâ€) is a mixture of TG, maltodextrin, gelatin, and an anti-caking agent. Gelatin is bonded extremely well by TG and therefore Activa GB forms strong bonds. It is not as versatile as RM because it cannot be made into a slurry, it is more sensitive to water, and it has a shorter working time. If you need a stronger bond, GB is useful.
- Activa GS: Â GS is the newest addition to the Activa line. It is nice because it can be left out on the counter all day without going bad. Like GB, it is a mixture of TG, maltodextrin, and gelatin, with added polyphosphate salts and a little oil. It is always used in the form of a slurry of 4 parts water to one part Activa GS. The polyphosphates make the slurry alkaline (basic, high pH). The enzyme is inactive at high pH, so the slurry is stable all day. Once the slurry is painted on meat, the pH drops, the enzymes become active, and gluing begins.
- Activa TI and TIU:TI is simply TG and maltodextrin with no added helper protein. TIU is the Kosher version of TI and is the only Kosher TG available. TI is packaged at twice the enzyme level as RM or GB. It can be sprinkled or made into a slurry. It will not bond as wide a range of foods as RM.
- Activa YG:YG a mixture of TG, lactose, maltodextrin, yeast extract, and safflower oil. It is designed to thicken and improve the texture of dairy systems like yogurt and cheese.
- Activa FP:FP is similar to RM but contains skim milk powder, which is friendlier on a nutrition label than caseinate (the useful part of skim milk powder). FP is useful for marketing a product that has an ingredients declaration.
IV. Storing and Testing TG
Ajinomoto ships Activa products in vacuum sealed foil pouches. Unopened, these pouches are good for 18 months if stored below 21°C (72°F). Once open, however, the enzymes become vulnerable to moisture. Opened pouches should be tightly wrapped and stored in the freezer. The freezing temperatures will not hurt the enzyme and the low humidity in the freezer will prevent degradation. Never leave large quantities of TG out on the counter, especially in open containers—they will quickly become garbage. Remove only the amount needed using clean, dry utensils and return the rest to the freezer. Keep the working container closed when not in use. A vacuum sealer is useful for breaking a batch of TG into small packages for sealed storage in the freezer.
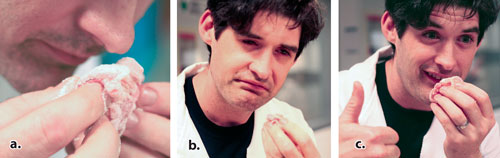
There is a way to test if your meat glue is still working. Get a small scrap of raw meat (we use chicken). Apply a liberal amount of meat glue to the meat and massage it in. Sniff the meat (don’t inhale the powder). If the meat smells like a wet dog or a wet wool sweater, your glue is good. If it doesn’t, your glue is bad. The next time you get a fresh shipment of TG, run the “wet dog†again and get a sense for how strong the smell is. After a while you will be able to tell how good your glue is (how high the enzymatic activity is) by how strong the wet dog smell is. Don’t wait too long to sniff after you massage in the glue because the smell dissipates after a couple of minutes. The wet dog smell is, I believe, caused by the small amount of ammonia released in the TG reaction. With one notable exception (see the above section on Safety Nutrition and Taste), I have never detected this smell in a finished product. The ammonia dissipates before you eat the product.
V. Salt and TG
Salt has an interesting effect on meats when combined with TG. Salting meat makes more of its proteins soluble and allows the meat to bind together naturally, as in a traditional sausage: Meat and salt are mixed together, the salt forces some of the meat’s protein into solution, and this protein forms a gel that binds the sausage together. In charcuterie this is called the primary bind. Because these soluble proteins are an excellent bonding substrate for TG, salting meat can dramatically increase bond strength. In many recipes, however, the combination of TG and salt can make food rubbery. Fix this problem by reducing the levels of salt or of TG.
VI. TG and Cooked and Cured Meats and Fats
TG depends on available proteins to do its job. Much of the protein on the surface of charred and seared meats is unavailable to TG for bonding. The Maillard reaction uses them up. The cooked portions of meats may or may not have the majority of its protein available. Typically, cooked meats will still glue together, but they do not bond as strongly as their raw counterparts. The added sodium caseinate in Activa RM (as opposed to Active TI) helps to overcome the lack of undisturbed proteins in cooked and cured meats. Bacon and ham can be glued, as can braised short ribs and other similar foods.
High fat items, like chicken skin and bacon, can be glued because of the connective tissue they contain. Rendered fat cannot be glued.
VII. Important Safety Note:
Â
Â
Â
VIII. Using TG
Activa RM is usually sprinkled over the pieces of meat to be bonded. Sprinkle enough RM on both surfaces to form a complete dusting; even out the coating with a brush. Alternatively, dip the pieces directly into the RM. RM can also be mixed with 4 parts by weight of water to form a slurry to paint on the meat. Don’t mix more slurry than will be used within 20 minutes, and discard the slurry after 30 minutes—the bonding power degrades over time because the caseinate in the RM will bonds to itself.
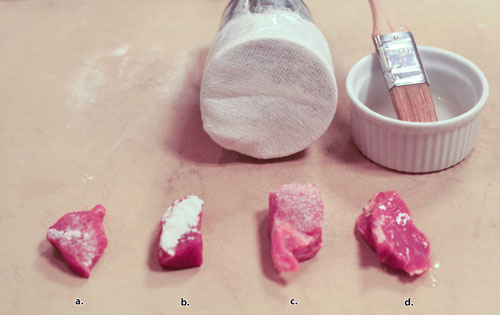
When bonding a mixture, add RM by weight. Typically, RM is used at .75% to 1% by weight of the mixture being bonded. 1 kilogram of mixture will require 10 grams of RM. The exception is pure chicken breast, which is more difficult to bond and sometimes requires up to 2%. Leg meat will work at 1%. Adding water or water-based flavors (like wine) to mixtures in small quantities does not affect bond strength, but adding fat-based flavors (like cream) will weaken the bond.
After adding TG, remove any air pockets between the two bonding surfaces. Vacuum pack food items to assure a tight seal, if the food and local regulations allow. For shapes like rolls and sausages, simply roll the products in plastic wrap. The plastic wrap technique is great because there are no rules regulating its use, and it is simple, fast and cheap; foods can be cooked in water directly in the plastic wrap. Putting a weight on top of pieces to be glued is also a good technique. Try to get all gluing and forming done within 20-30 minutes and then allow the product to remain undisturbed in the refrigerator for 4 hours or more, unless you are using the heat-set method.
TG will bond within 3 hours but bond strength will be substantially higher after 4 or 5 hours at refrigeration temperatures. Highly acidic environments (below pH 4) cannot be glued effectively. To make a TG ceviche, glue the seafood together before marinating in acid. Most other marinades, however, won’t lower the pH of the proteins enough to inhibit bonding, allowing for post-marinade gluing. TG, like most enzymes, increases its activity as the temperature is raised—to a point. The same bonding reaction that takes 4 hours in the refrigerator takes place in just 5-20 minutes at 50-58°C (122- 136°F). This technique is called heat setting. Typically we will place items to be heat set in a 55°C water bath. 5 minutes after the core has reached temperature the meat is bonded.
Â
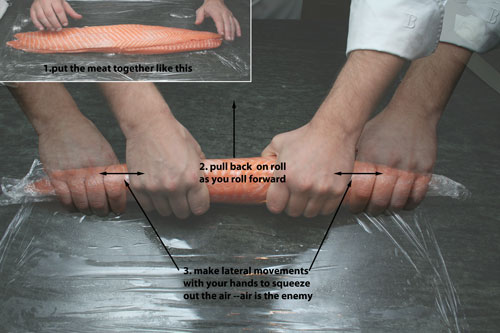
Rolling your own: More on the technique of rolling:
 If Nils had his way every animal in the world would grow in the shape of a tube. Tubes are great. Tubes cook more evenly and have less waste than other shapes. They can be cut into attractive and identical portions. TG gives a chef the ability to take odd meat shapes and make them into perfect tubes (or other shapes), producing consistently sized and shaped portions with minimum waste.
Sides of fish and beef tenderloins are notoriously difficult to use fully because of the thin tail sections. Here is how Nils rolls them. Pay attention because his technique is pretty good and it’s not as easy as it looks.



IX. A Note on Proteins:
Enzymes are proteins, and proteins are made up of building blocks called amino acids. Amino acids are small molecules that have three groups bonded to a central carbon atom—an amino group (meaning contains nitrogen), an organic acid group (COOH), and a side chain (usually referred to as the R group). There are 20 aminos that regularly occur in proteins and they are all the same except for the R group. It is the difference between R groups that give each amino acid its special characteristics. Some have a positive charge, some have a negative charge; some are bigger, some are smaller; some like water (hydrophilic) and some don’t (hydrophobic). All amino acids have the ability to be linked together by joining the acid group of one to the amino group of another. This link is called a peptide bond. TG does not form peptide bonds. TG bonds the R group of the amino acid glutamine to the R group of the amino acid lysine. Proteins are long chains of amino acids containing from a couple of hundred to several thousand units. The function of a protein is determined by its shape and the distribution of charge and hydrophilic and hydrophobic areas in that shape. These are determined by the order of the protein’s amino acids and the environment. Heating proteins makes them jiggle more violently, eventually disrupting their shape—the protein has been heat-denatured. Some denaturation is reversible and some, like cooking an egg white is not. PH (acidity and alkalinity) also changes the shape and function of proteins by altering the charge on the individual amino acids. This is why milk curdles when you add acid. Whipping and kneading can also change the shape and function of proteins—the unfolding of proteins in whipped egg whites allows us to make angel food cake.
In any enzyme reaction there is the substrate, which is what the enzyme reacts with, and the product, which is what the enzyme makes. The special shape of each enzyme determines what substrate(s) it will bind. It is a small section of the enzyme, called the active site, which actually does the work of catalyzing reactions. Enzyme function is often described using the lock and key model. The substrates fit like a key into the active site of the enzyme (the lock). When the enzyme is binding with its substrates the very specific geometry of the active site as well as distribution of charge and the placement of hydrophilic and hydrophobic groups within the active site allow reactions to occur very efficiently. If shape 24 of the enzyme is disrupted by heat, incorrect pH, etc, it ceases to function properly. It is important to know the heat and acid tolerance of a given enzyme when using it in the kitchen.
The speed of an enzyme reaction had many factors, including the inherent efficiency of the enzyme, the concentration of enzyme and substrate, the presence of inhibitors, and the temperature. As temperature rises, two things happen: first, molecules are more energetic, increasing the amount of available energy they have to react, and secondly, because molecules move faster when they are heated, they tend to collide more frequently, giving more chances for reactions to occur. In general, enzyme reactions will speed up as temperature increases until the enzyme is denatured. The heat denaturing of an enzyme is time and temperature dependant. Enzymes are typically stable up to a given temperature, which for TG is around 40°C (104°F), above which the enzyme is slowly inactivated. As temperature is increased, inactivation becomes faster and faster. Although is takes many hours to inactivate TG at 45°C (104°F), 10 minutes at 60°(104°F) will destroy 94% of TG.
If meat paste is extruded or quenelled into water or broth at this temperature, the outside sets almost instantly. Heat setting is the basis for Wylie Dufresne’s famous shrimp noodle dish. Dufresne mixes pure shrimp paste and salt with 1% Activa RM and squeezes the mixture directly from a Japanese noodle press into a water bath at 54.4°C (130°F), where it instantly sets into starch-less noodles. Any TG product can be set this way, although for food safety reasons choose a temperature below 54.4°C (130°F). Thin products will completely set in a few minutes; thicker products take longer. After setting, finish the product to its desired final cooking temperature. An immersion circulator is the best tool for this job, but pots of water on the stove will suffice in a pinch. The temperatures necessary for heat-setting TG will eventually inactivate the enzyme. Fortunately, the reaction is largely complete before the enzyme is destroyed. At 50°C (122°F) the enzyme loses a third of its potency after 10 minutes. At 60°C (140°F), it loses 94% of its activity after 10 minutes.
Excellent article. I can’t wait to play around with this stuff.
I want to wrap pork spare ribs in chicken skin, fry it up and make buffalo pig wings.
How much do you weigh?
we supply this kind of product,and could catch up step of Ajinomoto in case of our up to date R&D ability, if you like you can e-mail me or call me Tel: 0086-21-62848124
Fax: 0086-21-52844575
Mobile: 0086-13918831346
thanks for this! i took your hydrocolloids class last year and u guys gave us a crash course on TG. this is a perfect summary of that. i use the rolling technique all the time now. thanks dave thanks chef nils
brilliant
very well done! absolutely sums it all up in one place, rather than hunting and searching the web like others have had to.
Great writeup! When making casing-less sausages I assume that you would sprinkle the activa prior to processing to ensure even distribution?
Howdy Jake,
Sprinkle the TG into the mix just prior to rolling. We use it mainly for hand cut sausages where normal slating and mixing wouldn’t bind the meat sufficiently. IF you add TG to a normal sausage mixit can get very rubbery –either reduce salt (which reduces flavor), or reduce TG. As one of my Interns recently pointed out, you can take a standard sausage mix and dust the outside with TG just to give the outside of your caseless sausage more snap.
Awesome. Thanks for this.
I have finally managed to track down some TG so I should be trying it in the next few days.
Does the sodium caseinate in Active RM pose any threat to people with milk allergies?
Dunno. Think it’s pretty pure. If it’s a lactose problem I’d think you are Ok.
Hi Dave A,
I just really wonder how long have you studied TG?I felt a little shame when i was reading your excellent article.Because i’ve dipped into TG field for almost 1 year working in the Kinry Food Ingredients Co.,Ltd. I’m just a TG amature compared with you. :-)Our company are also original producer of TG with 10 years research .Please visit our website if you have interest. http://www.kinfi.com You know,Ajinomoto is first producer of TG in the world,actually,the transglutaminase is scale up by Amano of Japan and commercialized by Ajinomoto.We’re second one.
Do you have some information to share with us about TG application in the dairy products like yogurt and cheese?
Hi Webster,
Thanks for the compliment. I was introduced to TG by Wylie Dufresne soon after he started using it. Sometime in the early 2000’s. Nils and Wylie both learned about it at the same conference and were the first guys to use it in this country (that I know of). Nils used more TG at Aquavit than just about anybody (in the chef world). I havent done much with yogurt or cheese (except glue bacon to halloumi). I know some chefs doing dairy applications, like Johnny Iuzzini at Jean-Georges. He makes ricotta gnocchi with Activa YG. Can we try some of your product?
Hi Dave,
Please send me your mailing address by email.My email is zsw@kinfi.com
Our all Biobond (EB series) specially used for Restructuring muscle foods such red meat,poultry,seafood.
–Biobond EB3 works well as dry powder sprinkle.
–Biobond EB4 works well as water solution but is not intended for use as a dry sprinkle.
–Biobond EB(K) works well as dry powder sprinkle. Please don’t try water solution method because it’s kind of different to dissolve this type of TG powder in water.This Biobond EB(k) could achieve better binding effort than Activa RM.
Water Solution Type: Biobond EB4
Powder Sprinkle Type: Biobond EB3,EB(K)
The dosage we suggest for TG-EB3,EB4 is 0.5-2% ,for TG-EB(K),0.5-1%
Our Biobond WM, specially desighed to sausage and cooked ham for
1. Improved peelability and slice stability especially with “water thin†slices.
2. Reduced lossed during industrial slicing,less prduction brekdowns,increased slicing speed.
3. Inproved snap and firmness and reduced stickiness and chewinessl.
4. Standardization of material and process,no influence on yield.
and others Biobond TG products,like
Biobond S used for surimi product and other seafood
Biobond M for bakery and noodle
Biobond YG for yogurt
Biobond CH for cheese…..
You mentioned using TG at aquavit,what benefit could TG bring?
Most milk allergies are caused by pasteurization. The natural enzymes are destroyed by the heat. This is why pasteurized milk rots when left out. If you leave raw milk out it turned into something else (by these enzymes) that is still eatable . Try to find raw cow or goat milk. Raw milk is making a come-back. Goat milk is easier to digest then cow.
Why is it that we here in the USA consumes more dairy products then any other country! Yet we have the highest rates of osteoporosis (weak bones) ? Google it
Milk allergies are caused by lactose or casein proteins. so in answer it could be a problem for very sensetive individuals. As for mike if you leave raw milk out not only will it still cause allergic reactions, it will also be a veritable pertri dish of bacteria (I love unpasterised cheese but it is dangerous)
Do you know how much protein content a substance has to contain in order for the TGase to work? I was reading this article and started to think about it in ways other than bonding meat… do you think that spherification might work on a proteinous liquid, which would then allow you to sear the outside of it? Or maybe emerging a piece of soft cheese in it; it might form a skin that could stand up to heat but would burst open on contact with a fork unleashing a wave of cheesy goodness. Bread-less mozzarella sticks. Cheese bombs. Whatever I call them, I gotta get my hands on some of this stuff….
PS: Just found this page today. Its really something I’ve been looking for. Thank you.
PPS: People who are allergic to milk should not consume any form of casein (unless they’ve been tested and know for sure that it is a whey protein allergy.)
Hi Jimmy,
I don’t know how much protein you need. Obviously, the more protein, the stronger the result. I have never tried spherifying with TG. I assume you would use the same technique as methocel –hot oil (55C). The problem is that even at those temperatures the reaction will take place over several minutes. You have to make sure the balls don’t stick together. Hot water might also work. Wylie’s famous shrimp noodles are just TG shrimp and salt extruded into hot water. That is a paste, however. If your product is too liquid it won’t hold together. I think you’d have problems trying to do reverse spherification. If you dropped hot balls into a TG-Protein slurry, you could probably make a cross-linked coating, but you’d lose a lot of material because the pot life of the slurry would be short (20 min). It might be possible to make a Protein-TG-polyphosphate slurry that had an alkaline pH. That would be stable for hours. If the local conditions around the “hot balls” was acidic enough, you might get crosslinking at the surface of the ball. Dunno.
Where can I find this product in Canada
Dunno Chef Ian. You can order it from Dairy Land (I don’t know if they do Canada), or Le Sanctuaire in California (they ship to Canada). I don’t know if Terra Spice sells it. Have you tried direct contact with Ajifoods USA?
Chef Ian,you could try to contact with Thomas, Large & Singer INC which located in Markham, Ontario.For specific contact information,please make a desktop search.
Ultrafoods out of Milton, Ontario carries it.
Hi Dave,
Was wondering if Activa will bond pork belly since its high in fat content?
Thanks in advance!
Hi Mike,
Yes it will.
If the enzyme isn’t activated at a high PH, does this mean storage of the GS could be potentially longer? I understand that the phosphates are dry, but I’m assuming hydration is also a factor in the degradation of the enzyme?
Hi Justin,
I have wondered this also. I don’t know.
Thanks for the response Dave!
In regards to my question about the pork belly. Which TG should I use? GS/RM?
Also, I know you responded that “yes it will” to bonding the pork belly’s, but can I bond the fatty side of the pork belly to say, a non fatty side or even fat to fat?
Thanks in advance!
– Mike
Hi Mike,
I hope this reply works. Sometimes a reply to a reply gets lost. You can bond the fatty part of pork belly because it has never been rendered. You are basically bonding the protein based connective tissue (the stuff that would become the chicharron). It doesn’t bond as well as lean meat, but can be bonded. Rendered fat has no protein and can’t be bonded.
I would like to buy transglutaminase for meat.
How much is the price and what is the minimum order quantity and where could I get ?
my email address is ningrum28@yahoo.com
Hi Nunik,
We usually go through Ajinomoto or Le Sanctuaire to buy ours.
Nastassia
Le Sanctuaire has told me they no longer carry Activa. Anywhere where I can get less than a kg? Anyone want to share?
Hi Michael,
Did you try http://www.terraspice.com/index.php/terraspice/products/industrial_ingredients
Thanks Dave – just shot them an e-mail. Have you ever gotten from l’Epicerie? Seems like they are NY based as well?
Willpowder has it now as well.
where can I get the Activa TI or those this product have another name?
I think you have to get TI directly from ajinimoto
Hi there, can transglutaminase be used to glue vegetable together like how we used to glue meat?
There is a technique using tg to glue vegetables but it requires gelatin. See the forums for the thread on vegetable gluing using pectin.
If you vacuum-seal it immediately upon opening, will it survive long enough to be mailed somewhere? I’m thinking about splitting a package with some friends who live on the other side of the country.
I should think it would be OK.
I have a question: As a physician, we are seeing many people coming in complaining of abdominal pain, distention and diarrhea. The current thought is that some percentage of these people may have “gluten sensitivity”. We test for this by measuring an antibody produced against TRANSGLUTAMINASE. Is it possible that the skyrocketing rise in gluten sensitivity is due to the increasing use of this meat glue?
Hello Dr. Kroll,
I don’t think so. There is research that using TG might decrease the allergenic effects of gluten (but there is also research the other way). In any event, one of our crew is gluten intolerant (even a little does him a lot of damage) but he tolerates TG in meat quite well. I would recommend not using TG in gluten Free grain preparations until more is known, however.
quite possibly if eaten raw as transglutaminase in the body modifies gluten in several ways, the most common product is a covelant gliadin-transglutaminase protein that triggers auto-immune response (production of antibodies against transglutaminase) it would stand to reason that artificially produced transglutaminase would do the same. As the intolerance is genetic it would not cause intolerance in a normal person, but could theoretically increase sensitivity. The theory that it could reduce allergic reaction is possible if the reaction between artificial transglutaminase and gluten produced less covelant gliadin-transglutaminase protein than our bodies own transglutaminase does, Although this seems unlikley.
How much RM would it take to bond confit of thigh meat? Would it still be .75% to 1% ? Thank you so much!!!
I think less Tailroaster. If the confit pieces are quite small the level might go that high but for larger chunks you just need to ensure that the pieced are evenly coated and squished together tight with no air.
In the Harvard lectures, Wylie use TG and gelatin together to make a gel and barley that can be torched, but don’t melt. Do you know what the ratios are for gelatin to TG and what type of TG he is using
Hello Nathan,
Wylie uses Activa TI for that recipe (TG and Maltodextrin –no helper protein). He has said he thinks it would work with RM (which has casein) but I have had bad luck with it. My guess is the casein gets in the way of properly cross-linking the gelatin. I don’t know the ratio. I’m on the road now. Probably about a percent or so –mixed into the hydrated and slightly cooled gelatin before it is mixed into the dried out puree.
Amazing I could never find the research that supported my theory of the “off taste” from sous vide meats, I detected the off taste and a few others could not on short-ribs that were treated with activa RM and cooked for 72 hours. I never knew the explanation, thank you guys.
Rolled stuffed butterflied turkey breast in plastic film overnight. Got a great bond. Unwrapped and cold smoked it for 2 hours the next day- and the bond didn’t hold.
I was very surpprised- I’ve bonded turkey before with no problems.
Is smoke a problem with meat glue?
Any ideas?
Thanks,
Andrew
I can’t believe you ignored the risk of food poisoning. If pieces of steak beef are bound together with glue, then the bacteria that was on the outside of the original pieces will now be on the inside of the faux steak. If cooked rare, then there is a much greater risk of food poisoning. Why are you so irresponsible?
Apparently Mike, You missed the entire paragraph that I placed in boldface. Read carefully before you accuse. I will reproduce it again here:
VII. Important Safety Note:
Every chef knows not to fill a turkey with warm stuffing and put it in the refrigerator. Bacteria can multiply in the warm environment and produce dangerous toxins before the assembly gets cold enough to prevent such growth. TG presents the same kind of contamination risk. Do not glue warm food inside a product that will be stored. From a bacteriological safety standpoint, re-structured meats should be treated like traditional ground meat. When cooking rare, treat food like it will be consumed raw, as in a tartare. Rare cooking is acceptable on whole muscle cuts because the amount of bacteria found on the inside of muscles is low. Gluing meat puts the outside (with lots of bacteria) on the inside, so glued foods must be fresh and have a low bacteria count.
On a further note, I have noticed a recent spate of incorrect blog articles on Meat Glue recently. Some of the more egregious claims are:
Meat Glue is unsafe because of bacteria on the inside of meat. This is only true if that possible contamination isn’t taken into account. No one thinks a properly cooked hamburger is unsafe. Furthermore, meat can be cooked rare and still be pasteurized if the time and temperature curves are correct.
Meat glue has been banned in the EU. Rubbish. A type of protein bonder based on Thrombin was at issue, not the enzyme transglutaminase that is used by chefs.
Meat glue is used by chefs to rip off customers and glue “lots of tiny bits” into a “faux steak.” I spend most of my time railing against such poor uses of meat glue. That isn’t what we use it for.
Not sure why you advocate it when its banned in Europe ..and ..it has possibilities of offering food poisioning. possible contaminates once on the outside not locked inside whilst the unsuspecting griller eats his steak rare. How good is that?
see televised expose link below.
http://au.todaytonight.yahoo.com/article/8989315/consumer/meat-glue
S MItchell,
First off, I encourage everyone to watch the piece you linked to. It is rubbish. Transglutaminase isn’t banned in the EU (thrombin based meat glue is and that ruling was preposterous, the EU has a history of such nonsense). It is always illegal to glue up scraps and sell them as something they are not (a little something we call fraud). Furthermore, it is absurd to say “even an expert can’t tell” when meat has been glued together. Food poisoning is always possible with improperly handled products. I don’t know of any chefs that use TG to rip off their customers or butchers that sell reconstituted cuts to “unsuspecting grillers.” I’m sure they exist, but I don’t know them. Just because something can be used improperly doesn’t mean it shouldn’t be used. Might as well say you should never use water because someone might drown.
Do some more research and check the knee-jerk reactions.
Is TG kosher?
They used to make a kosher one, but I’ve never sourced it. Obviously Activa RM isn’t kosher, because it contains milk protein.
We are a original manufacturer of Transglutaminase (TG), which is a natural enzyme can cross link protein in the food and improve the product quality,it has been widely application. As a technology leading company focus on food enzyme, KFI could catch up step of Ajinomoto in case of our up to date R&D ability. By now, we have register our own brand ‘Biobond’ and have distributed in Russia, Thailand, Korea, Japan, Turkey, Austria, Argentina etc.We have many different formulas for different application to reach different results, which can use in meat, sausage, milk, wheat, soybean etc.And also we have certificates of Kosher, Halal and ISO. You can find detailed application of TG in our website: http://www.kinfi.com .
my address is s-support@kinfi.com
I encourage readers of this blog to go to sites such as the one above that linked to us and help squelch this knee-jerk unreasoned craziness.
Would you be willing to let us know if there are ever studies on TG being consumed the proper way (“down the hatch”) and not just via stomach pump? I’m concerned about long term use and proper use, as it has not really been studied. Even small changes like this to our diets can have horrible long term effects. We can see clearly that the more we alter things the more unhealthy we’ve become. The last 50-70 years have yielded some great culinary discoveries and have also lead us down a route of morbid obesity and a much lower overall condition of health. I’m concerned that this, like so many other things just hasn’t been looked into as it should. Also, is there any kind of documentation of this product or is the consumer just left to guess if it has been used in certain establishments?
Hello Carri,
At the time I wrote the primer on TG I read every article on safety I could get my hands on. If I remember correctly, it amounted to 500 or 600 pages (at least). My memory was that none of the studies were done by just having the animals eat the stuff (as opposed to using stomach tubes) –but that is just my memory, and there might be newer studies. I will say this, there were no studies I read, either pro or anti TG, that I didn’t discuss (I don’t have any hidden relevant data). Where the info seemed confused –as with coeliac disese and TG– I said so. When I found out that a study was funded by Ajinomoto, I said so, etc.
TG is a naturally occurring enzyme that isn’t “manufactured” in the way HFCS is –it the natural product of bacterial fermentation. To my knowledge, no GMO, no Frankenstein, etc. The only thing that separates microbial TG from the TG in your body (it is part of the formation of your skin and the blood clotting pathway, et) is that microbial TG is not calcium dependent.
As for your last question about documentation: a restaurant is not obliged to say it uses TG because it is not obliged to list the ingredients of its recipes the way a manufacturer is. On the other hand, if a restaurant says it is serving a “20 ounce ribeye” and they serve you a mess of scraps, that is fraud –and not allowed. There is a widely circulating Australian news clip that claims “not even an expert” can tell if something has been glued. This is patently false. Anyone who knows anything about meat can tell if a bunch of scraps are being foisted off as a whole muscle. One or two joints in a muscle look normal, and are the product of the proper use of TG –to make a cut better and more evenly cooked than the original. Numerous bonds look like a patchwork quilt –and don’t taste good either because in that scenario the texture probably won’t be right.
Can I use it to make Shrimp Steaks or Squid roasts ? This is exciting to me. Sometimes the only fish I can afford is the scraps can I wrap up two 3 different kinds of fish and grill these ? What about lobsters and craps, mussles,clams ?
Chuck Wow from Thailand.
Hello Chuck Wow,
You can bond either squid or shrimp into larger blocks of meat, but they won’t eat like a steak, they will eat like blocks of shrimp or squid. The trick when using TG is to respect the grain and structure of the meat as you bond such that the finished products texture makes sense and is delicious.
As for gluing scraps you will have several problems:
1. often, scraps don’t have the same texture as the whole muscle –gluing them together doesn’t help this
2. different fish have different tastes, textures, and cooking temperatures –sometimes they compliment each other, sometimes not
3. Fish fat tends to oxidize quickly, and scraps oxidize the quickest because of their unfavorable surface to volume ratio
4. Pieces of meat glues from scraps don’t taste like whole muscles –they are more like course cut sausages (when they are good) or a restructured nightmare crazy quilt (when they are bad)
5. (Most Important) Fish scraps are difficult to glue safely, unlike meat, which is relatively easy to pasteurize using proper low temp cooking techniques (for red meat cooked rare), or is normally pasteurized during cooking (pork chicken, etc). On fish, which you are unlikely to cook enough to kill bacteria in the center, you shouldn’t glue together anything you wouldn’t be willing to eat raw (which usually means no cheap scraps).
All in all, the best use for scraps is a delicious stew.
hey “chuck wow” if thats what you parents named you and you really live in thailand then you have my sympathies. Anyway I really live in thailand (Samut Prakan) do you know where to buy the stuff? i contacted ajinomoto but they didnt get back to me, thanks in advance
I am very gluten intolerant so my “beef” with this stuff is that I react very badly to it, but it doesnt have to be disclosed on any label. Even if a product is labeled gluten free, it still can have this stuff in it & cause me major issues. I hope that in the near future it will be law to disclose its use & much more research will be done on its effects on the gluten intolerant!
Howdy JML,
TG has to be labeled “enzymes,” so you should watch out for grain based products with that on the ingredient list.
I haven’t read the current research, but as my primer stated, some research points to TG aggravating gluten intolerance (presumably by making the small amount of gluten present in “gluten free” products more allergenic), whereas some research points to the possibility of reducing the aggressiveness of the allergy (presumably by bonding the protein to another in a way which makes it inactive). I tend to doubt TG will be helpful, however, because a known function of TG is to strengthen gluten formation in low protein flours (why would it make the gluten network physically stronger but allergically weaker, certainly possible, but not my first guess).
I hope that more research is done soon –I’m sure it will be because a lot of people care about the problem (so there is funding).
PS: I make chefs aware of the potential gluten problems when I teach.
More needs to be done to protect the public.
Certainly more research.
Definitely labeling.
In fact, I’d like to see a natural Antidote discovered to counteract these contaminants.
Chicken glued with Beef enzymes. Beef glued with Pork enzymes. Is nothing sacred anymore?
My husband is lactose intolerant…..sodium caseinate is no exception & he has experienced symptoms, even when no milk products have been consumed. So the culprit is now exposed.
I’m highly allergic to MSG…..which hides within a list of 100+ ingredients…..maltodextrin being one of them. Now I know why I continue to get ocular migraines & pass out for approx 3-5 hrs.
It’s debilitating. And the public has a right to know & choose. We can’t avoid what we don’t know our food is being bombarded with.
We will now avoid restaurant food. At home, we’ll eat meat on the bone. And we’ll continue to avoid meat saturated / injected with additives.
My motto: avoid anything adulterated by man.
Pat,
None of the TG enzymes we use are derived from meat sources –they are products of microbial fermentation. Activa contains no lactose (a milk sugar). Perhaps your husband also has a problem with milk proteins (casein). There is no MSG in Activa. Neither is there maltodextrin in MSG (it is a pure product). These products, as we use them, aren’t additives, and require no antidotes, for they are not poisons. Your comment seems more of a rant than a reasoned invitation to debate, which is what this blog likes to focus on.
Hi there. Can TG be used to bind 50% lamb+50% beef around 8kilo in weight minced and cooked on a spit.
Many thanks
Hi Rick,
Sure. Just be careful with salt. Let’s assume you would normally add salt and bind the mix into a sausage like consistency. If you did that AND added TG, the result would be bound too tightly –like a rubber ball. You have to reduce salt or reduce mixing so you don’t solubilize the native protein too much. Make sure the meat is cooked through to kill bacteria –but I’m guessing you already knew that.
Another incorrect bashing of TG. This blogger incorrectly conflates the pure enzyme TG with the product, Activa RM (whose active ingredient is TG). It would be the same mistake as saying we are taking way too much ibuprofen by confusing the maximum dose of ibuprofen in mg with the weight of the pills in grams. Read and see.
Thanks Dave.
I live in South East Asia. Where could i buy one kilo bags of Activa and what one would be best for the mix i am using.
Cheers Rick
Hmm. Dunno Rick, I’m sure Ajinomoto has a distributor in SEA. Here in the US they will usually give at least one free sample then sell direct in 1 Kilo bags.
Dr.Mercola sent an email out today May 4 2011 on just how dangerous this glue is. As an avid cook this stuff scares me, as a restaurant patron,this should be mentioned on the menus,so we have a choice whether we wan’t to consume transglutaminase aka meat glue or not. Does T. Keller & A.Waters use this ?
I have read the piece on the Dr. Mercola website and the information is horribly wrong. It rehashes the offensively poorly reported Australian piece on Meat Glue.
Dr. Mercola is an infamous quack. Turns out he’s a quack about food as well as medicine (no surprise)
see http://www.sciencebasedmedicine.org/?p=9903
Dr. Mercola (who, BTW, is not an MD) is a
quack. Among other things, he’s still making the thoroughly disproved case against childhood vaccines.
And JP, T.Keller’s cookbooks are filled with Transglutaminase recipes. I bought my first bag of the stuff to try a Keller recipe. Don’t know about Alice–my guess would be not.
As someone who avoids meat, doesn’t drink or smoke and is about 12 lbs overweight, I’m concerned about my diet because I had a heart attack three weeks ago!
I am neither diabetic nor do I have a cholesterol problem. In my case it’s genetic and stress.
And then I came across this:
http://articles.mercola.com/sites/articles/archive/2011/05/04/has-your-meat-been-glued-together–why-you-need-to-know-and-avoid-this-dangerous-process.aspx
As a Canadian I was wondering whether this “junk†is legal/available in Canada…and Googling it I found you.
Thought it might be of interest to you.
I have read it. It is incorrect.
yet another scaremongering fact less article throwing about statistics with no reference to sources. Why would an article trying to discourage using this product not mention the one health risk actually associated with it (gluten intolerance)?
You’ve got to be kidding. “THANKS” for your disservice, contributing to the complete craziness and scifi raping of Mother Earth – you are helping greedy companies completely destroy our food system and good natural health. You suck.
Amazing.
The ignorance of the replies in this story is just astounding.
Do these people also believe that the US Government was responsible for 911 and that Elvis shot JFK?
Loosen up those tinfoil hats people.
I use Activa to make a reverse bacon wrapped chicken and it works great.
Chicken breast skin removed and stuffed.
Wrapped with bacon, glued on.
Then skin glued on over the bacon.
Sear and roast. It’s delicious.
I think the Activa even has a tenderizing effect on the chicken.
Elvis didn’t shoot JFK? There goes another theory shot to hell.
How do we get Activa. It is not available in India.
Can any body help?
What?! This does not sound like something I want to celebrate. I would worry about the bacterial counts on the surfaces of the things you are gluing and then cooking rare. Also, glue derived from pork blood would render things non-kosher/halal and that should be taken into consideration for those who can’t eat pork. Ditto for the folks who don’t eat beef and a glue derived from cow blood is used. People not in the know about this stuff would never think that you would use pork or cow blood glue on something they want to eat. Besides, doesn’t it sound just a little bit gross?
Did you read my post before you wrote this comment? No one I know uses anything but mTG, which isn’t animal based, and we cook specifically to kill bacteria.
Just found this website because I’m trying to figure out a shampoo I bought that contains transglutaminase! Apparently it encourages curly hair to get curlier.
Wow. hadn’t hearda that.
Awesome! I am ordering more transglutaminase now. This is something I certainly want to try at home. I hope I don’t end up gluing my eyelids shut. I am a bit worried about clogging my shower drain, though.
I will grow out my hair, douse with transglutaminase, and go for the full on jew-fro.
Thanks, profnina!
Dave, your articles are informative and very readable. While I have heard a little about TG, you have opened a world of new information for me to explore. You suggested not gluing more then two or three pieces of meat together for stated reasons, would they apply if I wanted to make a tenderloin from medallions of pork, beef, and lamb. It would be a cylinder made of P|B|L|P|B|L medallions 1″ thick. Maybe even a scallop in the center. Do you forsee any issues arising from the TG?
Hello Saddler,
The TG itself will work. Since you are doing a special effect, the same rules need not apply. Pork beef and lamb can be effectively pasteurized, whereas scallop cannot –something to be aware of.
“Enzymes are proteins that act as catalysts in chemical reactions; they speed up reactions and make reactions occur that otherwise wouldn’t.”
Actually your wrong on that last part, enzymes cannot force a reaction that would not otherwise already occur naturally, each enzyme is reaction specific and works by lowering the required “activation energy”.
Overall I found your article to be quite informative, but I still don’t want to eat franken food made with this.
You are correct that they cannot “force” a reaction to occur that is energetically unfavorable, but they can make reactions occur that basically never would because the activation energy is too high, which is what I meant. It is energetically favored, for instance, for lysine and glutamine to bond together and release ammonia. In practice it won’t happen without the enzyme.
if you are using this meat glue you should be ashamed of your self, its terrible and can be hazardous to your health!
Really? Have you even read my post debunking this crazy video? https://cookingissues.com/2011/05/20/the-trials-of-transglutaminase%E2%80%94the-misunderstood-magic-of-meat-glue/
Dave,
If I’m going to be cooking something to above the 50c-58c threshhold for heat setting, say a deboned and rolled up half chicken, can I apply the TG, roll it up, vacuum seal it and go straight in to the circulator or will the higher temp deactivate the enzymes with out the overnight set?
Thanks
Howdy Chuck,
Typically, I put the item (like your chicken) into a bath at 55C and let it stay for a bit (20 minutes to an hour, depending on thickness), and then ramp up the circulator to the cooking temp.
I thank you for this site. I did not know that “meat glue” has been know of and used by chefs for so long. Although I am not in favor of using or ingesting this product (because of the many food allergies of my wife and I) I found your article informative and interesting.
Yes Ken, but the bond might not be quit as strong as with raw meat. The other problem is going to be getting your texture right. It is possible, but you might need to tweak your recipe around before you are satisfied.